HIV drug could
treat Alzheimer’s, age-associated disorders
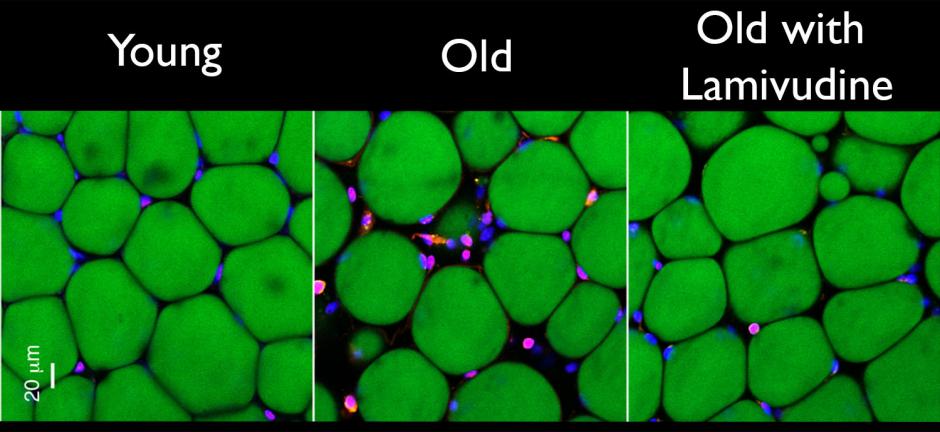
A new study found that an HIV drug significantly reduces age-related inflammation and other signs of aging in mice.
“This holds promise for treating
age-associated disorders including Alzheimer’s,” said John Sedivy, professor of
medical science and biology at Brown University. “And not just Alzheimer’s but
many other diseases: Type 2 diabetes, Parkinson’s, macular degeneration,
arthritis, all of these different things. That’s our goal.” Age-related
inflammation is an important component of age-associated disorders.
The findings were published on Feb. 6, in the journal Nature. The collaborative research project included researchers at Brown, New York University, the University of Rochester, Université de Montréal, the University of Virginia School of Medicine and Leiden University Medical Centre in The Netherlands.
According to Sedivy, the HIV drug
acts by halting retrotransposon activity in old cells. Retrotransposons — DNA
sequences able to replicate and move to other places — make up a substantial
fraction of the human genome.
Retrotransposons are related to ancient retroviruses that, when left unchecked, can produce DNA copies of themselves that can insert in other parts of a cell’s genome. Cells have evolved ways to keep these “jumping genes” under wraps, but as the cells age, the retrotransposons can escape this control, earlier research from Sedivy’s lab showed.
Retrotransposons are related to ancient retroviruses that, when left unchecked, can produce DNA copies of themselves that can insert in other parts of a cell’s genome. Cells have evolved ways to keep these “jumping genes” under wraps, but as the cells age, the retrotransposons can escape this control, earlier research from Sedivy’s lab showed.
In the Nature paper, the research
team showed that an important class of retrotransposons, called L1,
escaped from cellular control and began to replicate in both senescent human
cells — old cells that no longer divide — and old mice.
Retrotransposon replication, specifically the DNA copies of L1, is detected by an antiviral immune response, called the interferon response, and ultimately triggers inflammation in neighboring cells, the researchers found.
Retrotransposon replication, specifically the DNA copies of L1, is detected by an antiviral immune response, called the interferon response, and ultimately triggers inflammation in neighboring cells, the researchers found.
These retrotransposons are present
in every type of tissue, which makes them a compelling suspect for a unified
component of cellular aging, Sedivy said. Understanding that, the team
uncovered the interferon response, the potential mechanism through which these
jumping genes may cause cellular inflammation without necessarily causing
damage to the genome.
“This interferon response was a
complete game changer,” Sedivy said, noting that it is hard to track where newly
inserted transposable elements may have inserted themselves in a genome that
contains a vast number of inactive and active retrotransposon sequences.
The interferon-stimulating copies of
L1 DNA require a specific protein called reverse transcriptase. HIV and other
retroviruses also require reverse transcriptase proteins to replicate, Sedivy
said. In fact, AZT, the first drug developed to treat HIV/AIDS, halts HIV
reverse transcriptase.
Current multi-drug cocktails used to treat or prevent HIV/AIDS still contain specific reverse transcriptase inhibitors. Sedivy and his colleagues thought that this class of drugs may keep the viral-like L1 retrotransposon from replicating and thereby prevent the inflammatory immune response.
Current multi-drug cocktails used to treat or prevent HIV/AIDS still contain specific reverse transcriptase inhibitors. Sedivy and his colleagues thought that this class of drugs may keep the viral-like L1 retrotransposon from replicating and thereby prevent the inflammatory immune response.
The team tested six different
HIVreverse transcriptase inhibitors to see if they could block L1 activity and
the interferon response.One generic HIV drug, lamivudine,stood
out because of its activity and low side effects.
Growing human cells in the presence
of lamivudine did not impact when the cells reached senescence or kill the
senescent cells, Sedivy said. But lamivudine did decrease the interferon
response and the late-stage senescence-associated secretory phenotype (SASP) —
the important characteristics of senescent cells that promote inflammation in
their neighbors.
“When we started giving this HIV
drug to mice, we noticed they had these amazing anti-inflammatory effects,”
Sedivy said. “Our explanation is that although L1s are activated relatively
late in senescence, the interferon response reinforces the SASP response and is
responsible for age-associated inflammation.”
Treating 26-month-old mice (roughly
equivalent to 75-year-old humans) with lamivudine for as little as two weeks
reduced evidence of both the interferon response and inflammation. Treating
20-month-old mice with lamivudine for six months also reduced signs of fat and
muscle loss as well as kidney scarring.
The results were encouraging, Sedivy
said, but there’s more work to be done.
“If we treat with lamivudine, we
make a tangible dent in the interferon response and inflammation,” he said.
“But it doesn’t quite go back down to normal. We can fix part of the problem,
but we don’t actually understand the whole aging problem yet. The L1 reverse
transcripts are at least an important part of this mess.”
Sedivy is eager to translate the
findings to humans. Specifically, he would like begin clinical trials of
lamivudine for various age-associated conditions such as frailty, Alzheimer's
disease and arthritis.
Lamivudine was approved by the Food and Drug Administration in 1995, has been used to treat HIV/AIDS for decades, and its pharmacological activity and safety are well established, Sedivy said. The new clinical trials could be streamlined and focused on lamivudine’s efficacy in treating age-associated disorders, he added.
Lamivudine was approved by the Food and Drug Administration in 1995, has been used to treat HIV/AIDS for decades, and its pharmacological activity and safety are well established, Sedivy said. The new clinical trials could be streamlined and focused on lamivudine’s efficacy in treating age-associated disorders, he added.
He would also like to develop a
new reverse transcriptase inhibitor specifically for the L1reverse
transcriptase. To aid in developing a specific therapeutic with minimal side
effects, the molecular structure of theL1reverse transcriptase needs to be
determined, he added. Researchers could also develop other types of drugs that
target the L1 retrotransposons.
In addition to Sedivy, Marco De
Cecco, now an assistant professor (research) at Brown, was involved in the
research since its inception seven years ago. Other authors from Brown include
graduate students Takahiro Ito, Anna Petrashen, Amy Elias, Nicholas Skvir, and
Steven Criscione; visiting students from the University of Bologna Alberto
Caligiana and Greta Brocculi; and collaborating faculty Nicola
Neretti and Stephen
Helfand.
The National Institutes of Health
(grant P01 AG051449 and R37 AG016694 among others) funded the research.