Concerns remain about how drugs age on the shelf
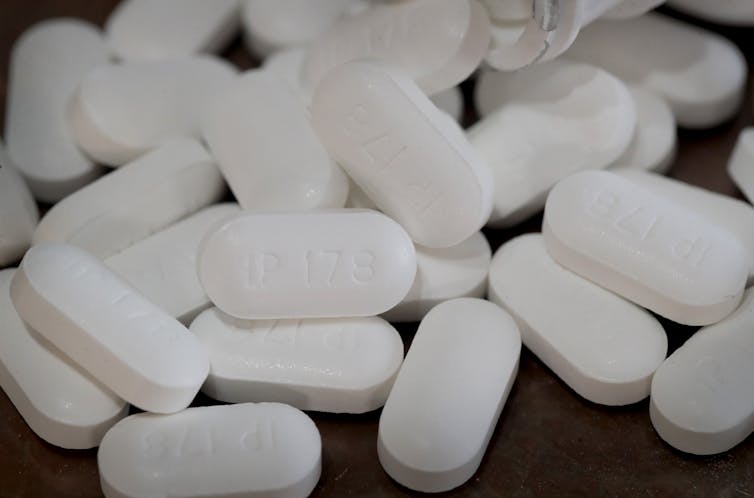 |
| Pharmaceutical companies recalled metformin, a Type 2 diabetes drug, after finding high levels of NDMA impurities. Scott Olson via Getty Images |
One dangerous result has been the creation of N-nitrosodimethylamine (NDMA), a probable carcinogen, in certain drugs. NDMA is found in chlorinated water, food and drugs in trace amounts.
To minimize exposure, the U.S. Food and Drug Administration has set an acceptable level of NDMA in each pill at less than 96 nanograms.
But over the past few years, the FDA has found excessive amounts of NDMA in several drugs for hypertension, diabetes and heartburn. As a result, the agency has initiated recalls to protect the public. These products were contaminated with NDMA during the manufacturing process. The FDA recommended best practices for manufacturers to minimize this risk going forward.
Unfortunately for the buying public, emerging evidence suggests that NDMA can also be created as some pills sit on the store shelf or medicine cabinet. Thus, there is no way to test for its presence in the factory.
I am a pharmacist who has written extensively about manufacturing issues and FDA oversight associated with both drugs and dietary supplements, including the issue of NDMA contamination. In January 2021, I discussed how NDMA can end up in a patient’s medication even if it wasn’t present during manufacturing. Two new FDA studies clarify how this chemistry works and may help partially ease patients’ concerns.
 |
| Zantac, the heartburn medicine, was pulled from the shelf, along with its generic versions, after the FDA found high levels of NDMA in the drug. Yichuan Cao/NurPhoto via Getty Images |
NDMA levels creep up after manufacture
Ranitidine (Zantac) was a commonly used heartburn and ulcer prescription and over-the-counter medication for decades before it was recalled by the FDA on April 1, 2020. It may now be the canary in the coal mine for the post-manufacturing creation of NDMA.
In one study, investigators found that ranitidine contained only 18 nanograms of NDMA after it was manufactured.
However, when stored at 158 degrees Fahrenheit (70 degrees Celcius) for 12 days – as if the drug had been left in a hot car – NDMA dosages rose above 140 ng. This is only slightly above the 96 ng limit the FDA has deemed safe, but this was after just 12 days.
In another study, storing ranitidine where it was exposed to higher temperatures or high humidity enhanced the creation of NDMA over time. This suggests that some medications can leave the factory with a safe amount of NDMA but if kept for too long at home or on the store shelf can exceed known acceptable limits by the time patients use them.
In a January 2021 study in JAMA Network Open, investigators simulated the stomach environment and found that when ranitidine was exposed to an acidic environment with a nitrite source, these chemicals could create more than 10,000 ng of NDMA.
However, FDA researchers more recently found that the earlier experiment used nitrite levels far beyond what would ever be found in the body. When they reran the study with normal dietary levels of nitrite, only a minimal amount of NDMA was created.
Further reassuring news came in May 2021, with the retraction of a clinical study from five years earlier. Originally, the researchers collected urine samples from 10 adults both before and after using ranitidine. After people swallowed ranitidine, their urinary NDMA levels rose from about 100 ng to more than 40,000 ng over the next day.
But now the scientists suggest that the NDMA levels they measured reflected what was created in the urine samples awaiting testing, rather than NDMA actually made inside the body. Furthermore, a new FDA trial that gave ranitidine to patients along with a high nitrite meal and did not find any concerning increases in urinary NDMA.
Other drugs need closer investigation
The body’s ability to create NDMA after taking ranitidine seems limited. But the fact that NDMA can be generated as ranitidine sits on the shelf or medicine cabinet is still concerning, and this same phenomenon might occur in other drugs as well.
In another study, investigators added chloramine, a disinfectant routinely added to sterilize drinking water, to water samples that contained one of several medications that are structurally similar to ranitidine.
They found that several commonly used drugs, including antihistamines (doxylamine and chlorpheniramine), a migraine drug (sumatriptan), another heartburn drug (nizatidine) and a blood pressure drug (diltiazem) all generated NDMA.
It is unclear whether the amount of NDMA created by these drugs when stored in hot and humid environments is dangerous, as with ranitidine. I believe that more studies need to be done right away to find out. It is always better to be safe than sorry, particularly when dealing with a possible carcinogen.
Editor’s note: This is an updated version of an article originally published on February 4, 2021.
[Deep knowledge, daily. Sign up for The Conversation’s newsletter.]![]()
C. Michael White, Distinguished Professor and Head of the Department of Pharmacy Practice, University of Connecticut and Adrian V. Hernandez, Associate Professor of Comparative Effectiveness and Outcomes Research, University of Connecticut
This article is republished from The Conversation under a Creative Commons license. Read the original article.